Home › Forum Online Discussion › General › Gone fishin'—for proteins (article)
- This topic has 0 replies, 1 voice, and was last updated 6 years, 1 month ago by
c_howdy.
-
AuthorPosts
-
December 23, 2019 at 1:47 pm #59592

DECEMBER 23, 2019
by University of Montreal
https://phys.org/news/2019-12-fishinfor-proteins.html
Using a new microscopic “fishing” technique, scientists at the Montreal Clinical Research Institute (IRCM) and Université de Montréal have successfully snagged thousands of proteins key to the formation of the cell skeleton.
Led by UdeM cell biologist Jean-François Côté, the team threw 56 “baits” into human cells they were incubating in their laboratory, catching more than 9,000 proteins in the process. The results are published today in the journal Nature Cell Biology.
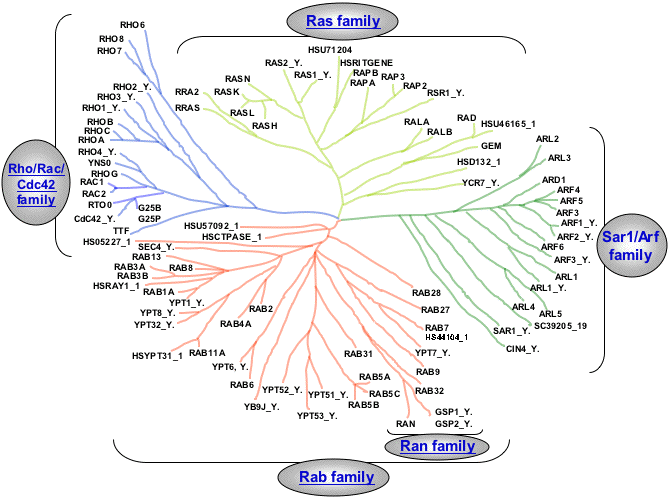
The goal was to identify the proteins that attach to those of the Rho family, famous in the cell biology world since the discovery in the early 1990s that they dictate how pieces of the cell skeleton—the “cytoskeleton”—are assembled.
In humans, the 20 members of the Rho family are scattered on the inner surface of cell membranes and act like small switches. When a signal from outside or inside the cell activates them, they stimulate other proteins to force the cytoskeleton to add or remove parts to its framework.
Out of all these proteins, only three, to date, have been thoroughly studied by researchers: Cdc42, Rac1 and RhoA. Cdc42 acts as the lead protein: it indicates the path that white blood cells must take to find a site of infection. Rac1 activates the engines that drive a non-muscular cell forward. RhoA stimulates the formation of fibres that allow cells to contract or form resistant tissues as they come together to produce, for example, the wall of a blood vessel.
But what are the other proteins doing? And what other proteins do they interact with? To find out, Coté and his team went fishing for answers.
Into human cells growing in incubators in their lab, they cast their baited lines, forcing these cells to produce proteins with two heads, one containing a single side of a protein in the Rho family, the other a “biotin ligase” enzyme. The latter acts like an elite sniper in the cell, luring and labelling every passing protein with the help of its partner, a member of the Rho family. Every protein that approached the bait was thus labeled with biotin. Next, Côté and his team had to shred the cells to identify, one by one, each labelled protein.
Using 28 two-headed proteins and presenting the GTPases—a superfamily of enzymes that function as ‘molecular switches’ and are involved in regulating many cellular processes—in both active and inactive configurations, the team caught a total of 9,939 proteins. Some were already known to the scientists, including the GTPases’ activators and deactivators. But the researchers also discovered hundreds of individual proteins with yet-to-be-defined roles.
These discoveries include the missing link of the cytoskelton Rho process identified in the early ’90s. Back then, researchers noticed that the RhoA protein indirectly activates another protein, ERM, causing it to stabilize the cytoskeleton. But they didn’t know the precise mechanism behind this process. On their “fishing expedition,” Côté and his team found the answer: what forges the link between RhoA and ERM is a protein called SLK.
In their study, the IRCM team also looked at other proteins that, until now, were virtually unknown to biologists, namely GARRE and PLEKHG3. The scientists demonstrated that these proteins naturally attach to the active forms of Rac1 and RhoG, respectively. What’s left to understand is the exact function of these associations. To accelerate the process, the team revealed characteristics of other molecules they caught in their experiments—enough to give raw material to dozens of laboratories worldwide.
Through their research, Côté and his team have not only cleared up a whole area of cell biology, but have also demonstrated the effectiveness of their unique “fishing” method. Côté now plans to use it to better understand how other molecular switches work, especially those in the Ras family, proteins that lie at the centre of many types of cancer.
“Mapping the proximity interaction network of the Rho-family GTPases reveals signalling pathways and regulatory mechanisms,” by Jean-François Côté et al, was published Dec. 23, 2019 in Nature Cell Biology.
More information: Mapping the proximity interaction network of the Rho-family GTPases reveals signalling pathways and regulatory mechanisms, Nature Cell Biology (2019). DOI: 10.1038/s41556-019-0438-7 , https://nature.com/articles/s41556-019-0438-7
Journal information: Nature Cell Biology

-
AuthorPosts
You must be logged in to reply to this topic.
